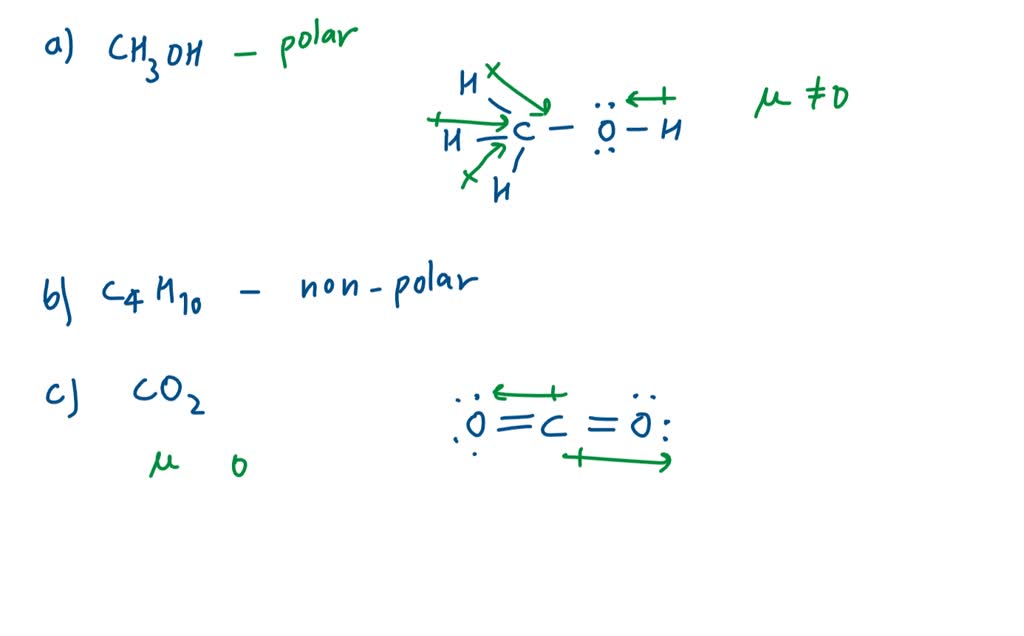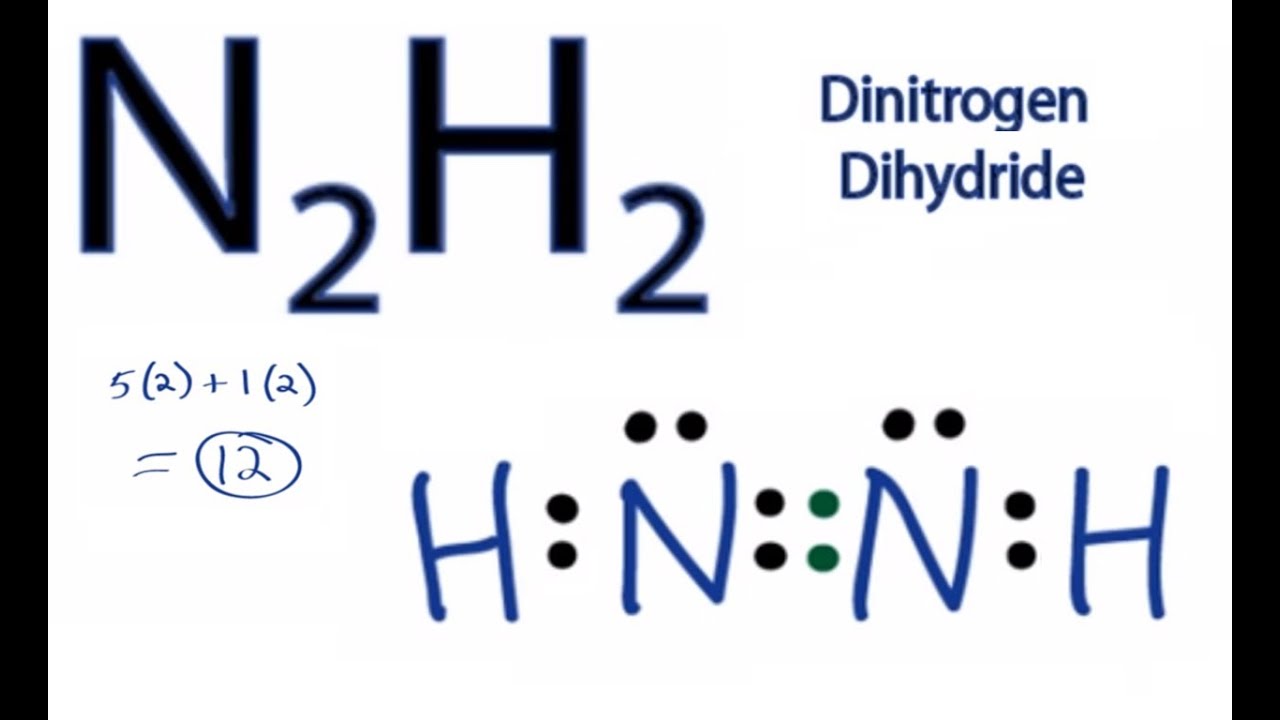Draw the molecules. Include all lone pairs of electrons. N2H2 N2H4 C2H2 C2H4 H3COCH3 | Homework.Study.com

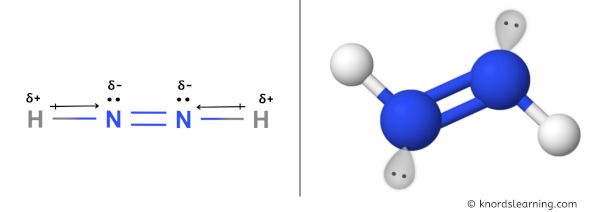
N2H2 Molecular Geometry, Bond Angles & Electron Geometry (Diimide) | N2H2 Molecular Geometry, Bond Angles & Electron Geometry (Diimide) Molecules like N2H2 can be a little tricky because although they might seem
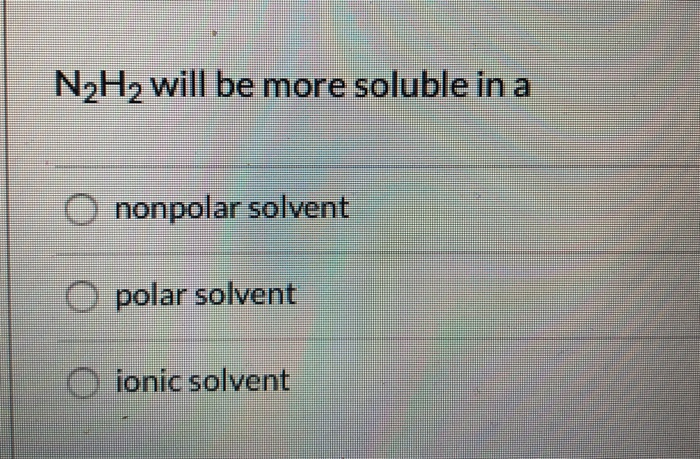
N2H2 is polar molecule with london dispersion forces,dipole dipole forces and H bonding .True or false?How?